
Microplastics and Health: Unraveling the Science with Dr. Samantha Hughes
Plastic pollution stands as one of the most urgent environmental crises of our era, yet its full impact on human health, wildlife, and ecosystems remains elusive. Microplastics, in particular, have ignited fierce debate—some studies sound the alarm over catastrophic consequences, while others suggest their effects may be overstated.
To cut through the noise and dive into the science, I reached out to my longtime friend, Dr. Samantha Hughes, an assistant professor at Vrije Universiteit Amsterdam (VU), a global leader in environmental toxicology research. Dr. Hughes has established herself within the group, with some of her latest work focusing on microplastics and the endocrine-disrupting chemicals found in plastics. In this conversation, we unpack what science knows—and doesn’t yet know—about microplastics’ health effects. We also explore how a tiny but mighty organism, Caenorhabditis elegans (C. elegans), is shedding light on the biological toll of these pervasive pollutants.
Yuliana: Sam, can you tell us about your role and what you’re currently working on at Vrije Universiteit Amsterdam?
Dr. Hughes: I’m an assistant professor in the Environmental Health and Toxicology department at VU. Our group uses a variety of model systems—cell cultures, organoids, zebrafish embryos, and my personal favorite, the nematode C. elegans—to investigate how environmental contaminants affect human health. My research focuses on chemicals like pesticides, endocrine-disrupting chemicals (EDCs), and per- and polyfluoroalkyl substances (PFAS), exploring their effects on growth, development, and reproduction. A key interest of mine is transgenerational impacts—how exposure today might ripple through generations, affecting not just individuals but their children, grandchildren, and beyond.
Lately, I’ve expanded into neurodegenerative diseases like Parkinson’s. We’ve built a C. elegans-based platform to model Parkinson’s and test how pharmaceuticals influence its progression. This work is evolving to investigate how pesticides may influence the development and progression of neurodegenerative disease.
Yuliana: What makes C. elegans such a great fit for toxicology research, and how well do the findings translate to humans?
Dr. Hughes: C. elegans is a tiny, 1mm-long nematode that’s surprisingly human-like in many ways. It shares fundamental processes with us—growth, reproduction, metabolism, even programmed cell death (apoptosis)—all of which we can study up close. It also has nerve cells akin to those in mammals, though it doesn’t use adrenaline.
Beyond biology, C. elegans offers practical perks. These worms are self-fertilizing hermaphrodites, so a single worm can produce up to 300 genetically identical offspring. Their life cycle is lightning-fast—just three days from egg to adult—and they live for three to four weeks, letting us study all life stages easily. Plus, under the relevant legal and ethical frameworks for research, they’re not classified as “animals,” so we don’t need special approval to work with them.
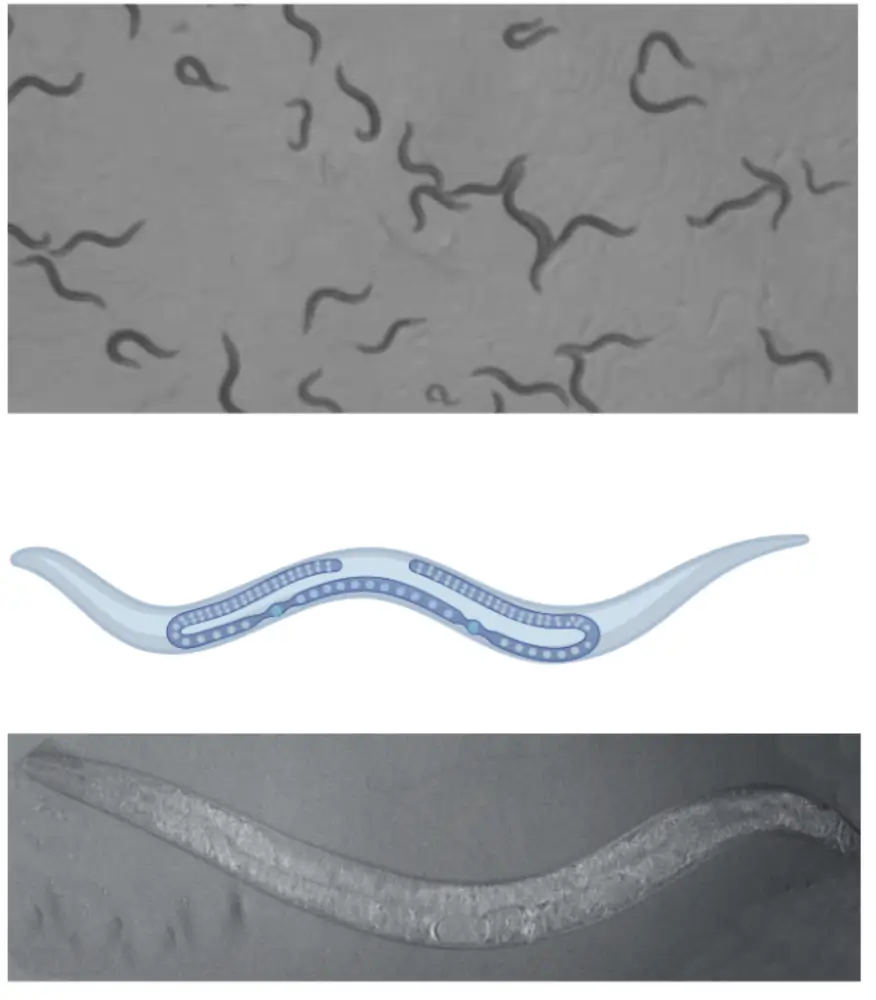
We use C. elegans to track how chemicals affect their development, survival, reproduction, and movement. It’s an ideal starting point for understanding toxicity in living organisms. That said, there are limits. For example, worms do not have the complex immune system that humans have, which plays a big role in how mammals respond to toxins. There are also differences in how toxins enter the body and are processed. So, while C. elegans offers critical clues about potential human health risks, we need follow-up studies in more complex models to confirm those insights.
Yuliana: Right, it’s fascinating how much we can learn from these tiny worms, but it’s premature to extrapolate findings directly to humans, assuming a 1-to-1 relationship—despite what some flashy headlines might suggest. That brings me to a topic that’s been grabbing plenty of those headlines lately: microplastics. They’ve also become a key focus in your recent work. What’s driving this sudden surge of interest?
Dr. Hughes: The surge in attention comes from a growing realization of just how massive and pervasive plastic pollution has become. Microplastics—tiny fragments from broken-down larger plastics—are now everywhere: soil, water, air, and even our bodies. At VU, our Environmental Health and Toxicology team, alongside collaborators, has detected microplastics in human blood and placental tissue. That discovery raises urgent questions about what these particles might mean for our health. The next big challenge is figuring out how they accumulate in organisms and what that buildup does.
Yuliana: We’ve seen microplastics being detected in a growing number of human tissues, but the health impacts still seem murky. What do we know so far, and why is it so hard to study this in humans?
Dr. Hughes: You’re right—the health effects are still a big question mark. Microplastics show up in organs and tissues, but we don’t yet know what that means for us. One hurdle is their diversity—size, shape, and chemical makeup vary wildly, making it tough to pin down universal effects. Another issue is timing: symptoms from exposure might take years to surface in humans. Then there’s the classic correlation-versus-causation dilemma. When we detect microplastics in diseased human tissue, are they the ones causing the pathology, or are they just more likely to pile up in already-damaged tissues? We can’t say for sure yet.
That said, plastics often contain additives, such as plasticizers and flame retardants which can leach out from them, especially when exposed to environmental stressors such as heat and UV light. Some of the additives – bisphenol A (colloquially known as BPA) is a big one – are known to be endocrine disrupting chemicals, which can interfere with and prevent the binding of natural hormones to their receptors and/or act as hormone mimics, thus affecting the normal function of the natural hormones produced by our bodies.
We also have to consider the effects of the physical presence of microplastics, which are essentially foreign bodies in the human system. As such, when microplastics enter the body, they could trigger an inflammatory response. Microplastics can potentially also physically damage the cell leading to stress and inflammation.
The complexity—diverse particles, slow-emerging effects, potential accumulation, and variable exposure—makes this a tough nut to crack. At VU and beyond, we’re working to untangle cause and effect, but it’s a slow process.
Yuliana: How does C. elegans fit into this puzzle?
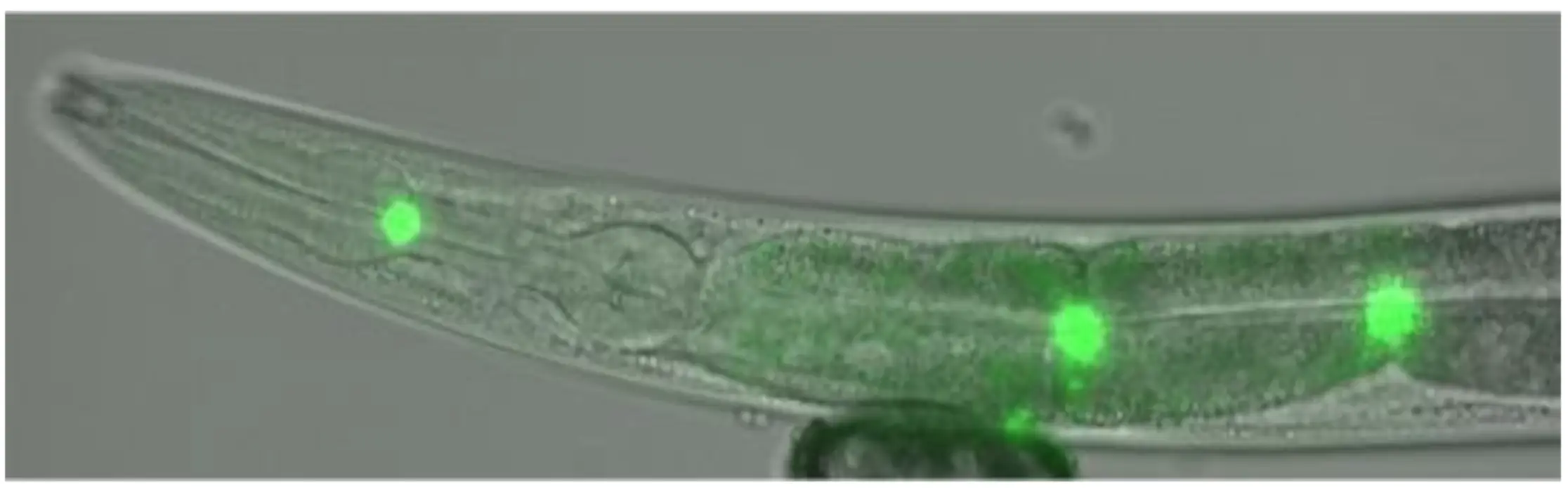
Dr. Hughes: C. elegans is a game-changer here. It provides a controlled environment where we can precisely test how microplastics affect a living organism, adjusting factors like plastic type, size, concentration, and exposure duration. The worms actually eat the microplastics, and by tagging the particles with fluorescent markers, we can watch them travel through the gut (see image below) and see if they spread to other tissues or even offspring. We can also measure how microplastics affect growth, reproduction, behavior, and survival. It’s a foundational step to understanding health risks that would be much trickier to study directly in humans or larger animals.
Yuliana: What’s next for your microplastics research?
Dr. Hughes: I’m collaborating with a brilliant PhD student at VU to explore how microplastics impact reproduction and development. We’re wrapping up a paper on it now—stay tuned!
Yuliana: Can’t wait to read it! Thanks, Sam, for breaking this down for us.
Additional resources:
Learn more about the University of Vrije’s toxicology research here. Some of their most recent studies in the field are highlighted below.
van Boxel et al. (2024) Effects of polystyrene micro- and nanoplastics on androgen- and estrogen receptor activity and steroidogenesis in vitro. Toxicology in vitro 101:105938
Brits et al. (2024)Quantification of micro and nanoplastics in human blood by pyrolysis-gas chromatography-mass spectrometry. Microplastics and Nanoplastics 4(1):12
Leslie et al. (2022)Discovery and quantification of plastic particle pollution in human blood. Environment International 163:107199
Dr. Samantha Hughes’ bio

Samantha Hughes is an Assistant Professor at the Environmental Health & Toxicology Department at the Vrije Universiteit Amsterdam. Her research focus is on how we can live longer and healthier lives. She studies the impact of chemicals on health outcomes using the nematode worm Caenorhabditis elegans as a model organism. Before starting at the VUA in August 2021, Samantha was a Principal Investigator at the HAN University of Applied Sciences, where she worked closely with colleagues in academia and industry to identify new healthy foods and drugs to treat neurodegenerative disease. Before moving to the Netherlands in 2016, Samantha did her postdoctoral research at the University of Oxford (UK) using roundworms and flatworms to explore cancer pathways. Before starting her postdoc, Samantha obtained her PhD at Cardiff University (Wales, UK) and King’s College London (England, UK) and a BSc degree in Biochemistry at Cardiff University.
 TM
TM
 TM
TM
We are a team of molecular biologists, geneticists, engineers, and data scientists brought together by the desire to solve difficult problems with far-reaching implications.
Address
423 Hindry Ave, Suite B, Inglewood, CA 90301, United States
© 2025 All Rights Reserved. Website by Astrael


